Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 25.06.2024

In classical solid phase peptide synthesis, typically, at least two reaction steps – namely deprotection and coupling – have to be carried out to grow a peptide chain by one amino acid. Thus, the synthesis of a 10mer requires already 20 steps. Assuming that the yield per step is 98%, the yield of the target sequence is only 67%. Nevertheless, within a longer sequence, the probability that difficult fragments are appearing is rather high, which will lower the overall yield of the synthesis even further. In contrast, an average yield of 99.9% per coupling step will result in 98% global yield.
The yields per step depends on the efficiency of coupling and deprotection. Even if general protocols exist, method optimization and process controls are highly recommended, especially for difficult sequences or in the case of upscaling. In addition, to avoid deletion sequences and simplify purification, capping after each coupling step should be considered.
Amino-deprotection can be monitored by Fmoc release and coupling with the activated carboxy group of the added amino acid by disappearance of the free amino group.
As long as no interfering reagents like, e.g., HOBt or Oxyma are used, the release of fluorene upon deprotection with piperidine may be directly followed by UV/Vis spectroscopy, even online with a flow-cell. Upon treatment of the Fmoc-protected amino acid with piperidine (20% v/v) in DMF, deprotection occurs and the resulting piperidine-dibenzofulvene adduct can be quantified at 289.9 nm or 301.0 nm (ε = 7100-8100 l/mol/cm; various values are reported in the literature) using Lambert-Beer’s Law. This method may also be used to estimate the loading capacity of the resin, an important parameter in peptide synthesis. For reliable results, 10 to 20 mg of resin should be used. On-line monitoring of the coupling and deprotection reactions also can be achieved by continuously measuring the refractive index of the liquid phase.
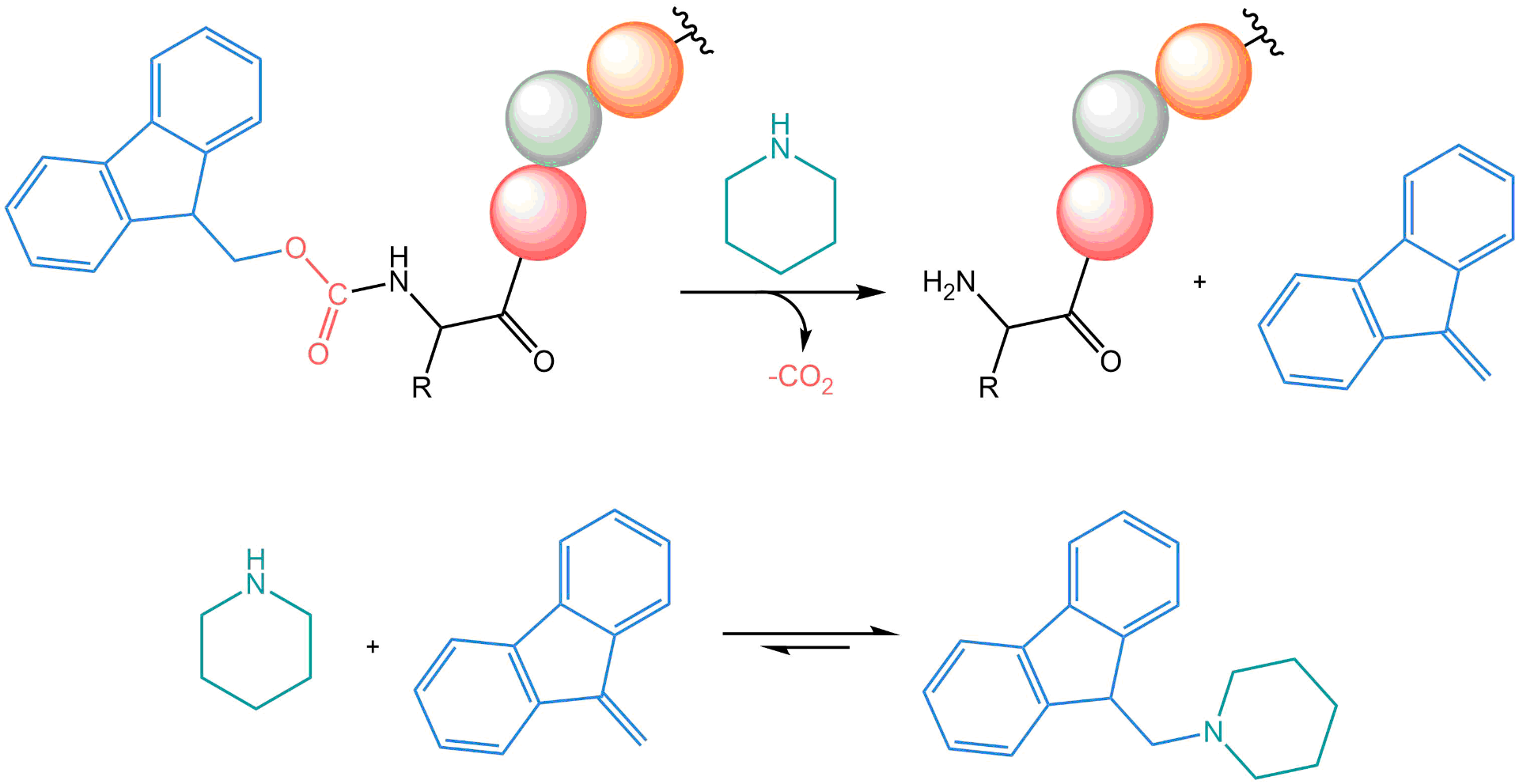
Piperidine is used for Fmoc protection; the adduct of the released dibenzofulvene and excess piperidine can be quantified spectrophotometrically.
Another possibility of reaction monitoring is to sample aliquots of the resin from the reaction vessel, to stain the accessible amino groups, and to analyze the beads under a microscope. There are qualitative and quantitative testing procedures. However, these are inconvenient for the use with automated peptide synthesizers and rather used for the optimization of large-scale processes.
Besides, primary and secondary amines, esp. of amino acids, can be visualized by ninhydrin staining. This very sensitive test has been adapted for SPPS and is known as Kaiser Test: A few resin beads are sampled from the coupling reaction, washed, and then treated with a few drops of each of these solutions: 50 mg/ml ninhydrin in ethanol, 1 g phenol in 0.25ml ethanol, and 2% (v/v) 1 mM potassium cyanide in pyridine. After heating for 5 min at 100 °C in an oven, the resulting color is evaluated: If free amino groups have been present, the resulting color will be blue to violet, if no free amino groups have been present, the color of the reaction products is yellow. In this way, SPPS may be monitored for completeness of amino-deprotection (blueish color), as well as coupling (complete: yellow color, absence of a blueish tint). In case incomplete reactions are noticed, deprotection and/or coupling times need to be extended or the reaction step needs to be repeated. However, N-terminal Asp, Asn, Cys, Ser, and Thr yield rather unspecific colors and N-terminal Pro will give brownish-red results.
Another chromogenic assay for primary amino groups is the so-called TNBS-Test (TNBS = 2,4,6-trinitrobenzenesulfonic acid), which is specially suited for the microscopic evaluation of resin beads. The completeness of the coupling reaction may easily be followed by assessing the homogeneity of the beads’ coloration. An orange color of otherwise colorless and clear beads indicates free amines and means incomplete coupling. For the TNBS-Test, a few beads are reacted with a few drops each of 10% v/v diisopropylethylamine (DIPEA) in DMF, and 1M TNBS in water.
For the staining of secondary amines like prolines, the acetaldehyde/chloranil test may be used. Therefore, a few beads of the reaction are treated with 2% v/v acetaldehyde in DMF and 2% v/v chloranil (tetrachloro-1,4-benzoquinone) in DMF. After incubating for 5 min at ambient temperature, the beads are examined under the microscope. Secondary amines produce dark blue to green beads. If the beads are colorless to yellowish, no secondary amines were present.
For all tests, we recommend the use of reference materials, i.e., materials of known reaction status, like samples which were drawn before a reaction step is performed or which were extensively treated to ensure complete conversion.
References:
High-Efficiency Solid Phase Peptide Synthesis (HE-SPPS); J. M. Collins, K. A. Porter, S. K. Singh, G. S. Vanier; Org. Lett. 2014, 16(3): 940-943. https://doi.org/10.1021/ol4036825
Color test for detection of free terminal amino groups in the solid-phase synthesis of peptides; E. Kaiser, R. L. Colescott, C. D. Bossinger, P. I. Cook. Anal. Biochem. 1970, 34(2): 595-598. https://doi.org/10.1016/0003-2697(70)90146-6
Substitution determination of Fmoc-substituted resins at different wavelengths; S. Eissler, M. Kley, D. Bächle, G. Loidl, T. Meier, D. Samson; J. Pept. Sci. 2017, 23(10): 757-762. https://doi.org/10.1002/psc.3021
Advances in Fmoc solid-phase peptide synthesis; R. Behrendt, P. White, J. Offer; J. Pept. Sci. 2016, 22(1): 4-27. https://doi.org/10.1002/psc.2836
Refractive Index: The Ultimate Tool for Real-Time Monitoring of Solid-Phase Peptide Synthesis. Greening the Process; B. de la Torre, S. Ramkisson, F. Albericio, J. Lopez; Org. Process. Res. Dev. 2021; 25(4): 1047-1053. https://doi.org/10.1021/acs.oprd.1c00051