Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 19/07/2016
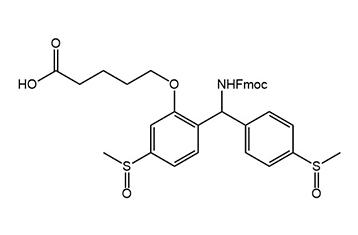
The correct choice of linker is pivotal for the success of a solid-phase synthesis. Commonly, linkers for solid-phase peptide synthesis are cleaved under acidic or basic conditions. This usually limits the possible protecting group strategies to either the Fmoc or the Boc strategy, respectively. Also the choice of side-chain protecting groups is restricted.
Iris Biotech now offers the innovative Safety-Catch Acid-Labile (SCAL) linker, using the 4,4'-dimethylsulfinylbenzhydryl (Msbh) moiety as cleavable element. The SCAL linker is, until being activated by reduction, stable to all but the harshest acidic conditions, as well as many basic conditions. Furthermore, the SCAL linker shows stability towards some reductive conditions such as hydrostannolysis, or Alloc deprotection conditions.
Consequently, SPPS on a solid support equipped with the SCAL linker can be performed by using the Fmoc-, the Boc- or the Alloc-strategy.
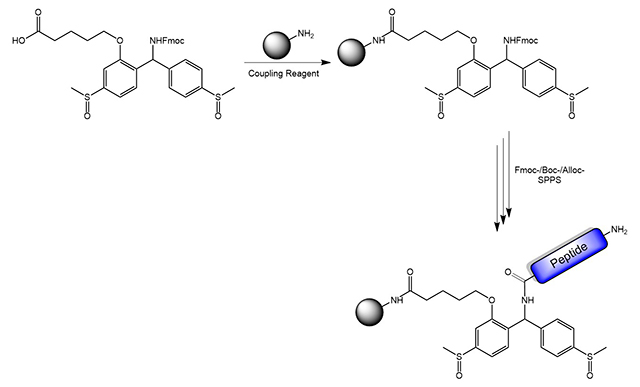
Cleavage can occur either by using a two-step procedure of reduction and subsequent acidolysis (shown below), or by a one-pot reductive acidolysis (e.g. using SiCl4/anisole/TFA). Cleavage of the peptide from the solid support affords a C-terminal peptide amide.