Welcome to Iris Biotech
For better service please confirm your country and language we detected.

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 25.07.2023

In nature, the CBT-Cys click reaction is reported as part of the D-luciferin regeneration pathway in bioluminescent animals, e.g., fireflies, where 5-hydroxy-2-cyanobenzotriazole (2-CBT) and predominantly D-cysteine are forming D-luciferin, a light generating substrate for the enzyme luciferase.
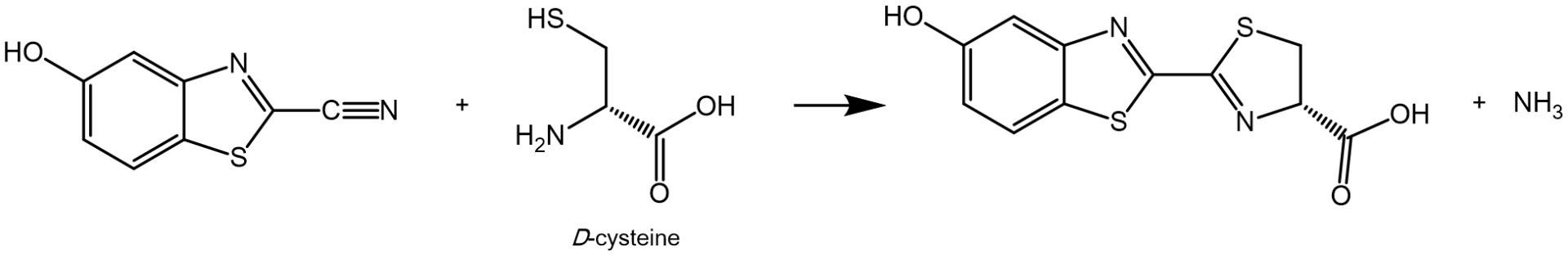
Biosynthesis of luciferin from hydroxy-2-cyanobezotriazole and D-cysteine.
In the biomimetic CBT click reaction, the nitrile function of 2-cyanobenzothiazole (2-CBT) reacts with a beta-aminothiol (1,2 aminothiol) and forms a 4,5-dihydro-1,3 thiazole. The beta-aminothiol may originate from, e.g., an N-terminal D- or L-cysteine residue, which can be easily introduced recombinantly or during SPPS.
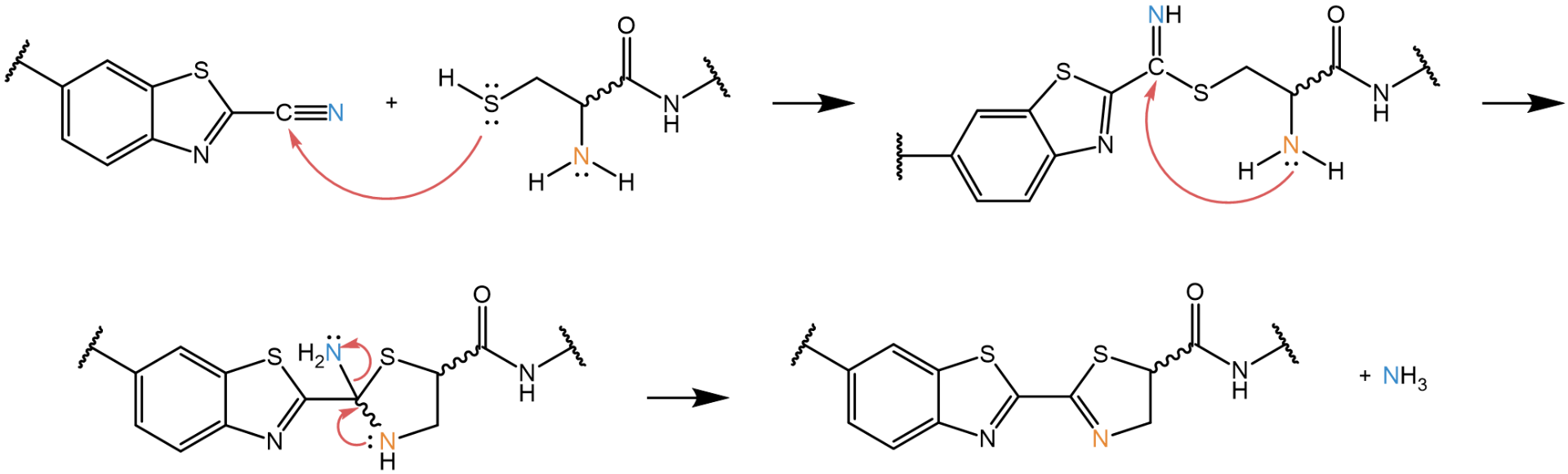
Mechanism of the CBT-Click reaction: The electron pair of the sulfur attacks the carbon of the cyano group, the nitrogen forms an enamine. The cysteine’s nitrogen then attacks the carbon of the enamine, and a cyclic intermediate is formed. Finally, the lone electron pair of the amino nitrogen of the cysteine attacks the positively charged carbon to yield the product. The nitrogen which came from the nitrile is leaving the reaction as ammonia.
Especially for biochemical and biomedical applications, the CBT-Cys click reaction bears several benefits: it is highly efficient at physiological conditions, about 120 times faster than the copper mediated azide-alkyne click reaction (CuAAC), and does not require catalysis by potentially toxic heavy metal ions (copper or ruthenium). The reaction may be used, e.g., to functionalize biomolecules, to add payloads, to fuse peptide fragments, or to generate sophisticated probes for in vitro and in vivo imaging.
Besides utilizing an N-terminal cysteine, a beta-aminothiol can also be introduced within the peptide sequence via a side chain modification, e.g., via a deliberately protected lysine. An economical way is to incorporate Lys as Fmoc-L-Lys(Mmt)-OH (FAA1622), which may be deprotected with 1% TFA as final step of the peptide synthesis. For other lysins, Boc (which requires higher TFA concentrations for removal) is reported for the side chain protection (FAA1125). Then, Fmoc-Cys(Mmt)-OH - available as L- (FAA1030) or D-isomer (FAA1614) - can be attached to the side chain of that lysine. The Fmoc is removed from Cys with piperidine in the final deprotection step, together with the N‑terminal Fmoc, and the Mmt residue is removed during the recovery of the peptide from the resin with TFA in DCM.
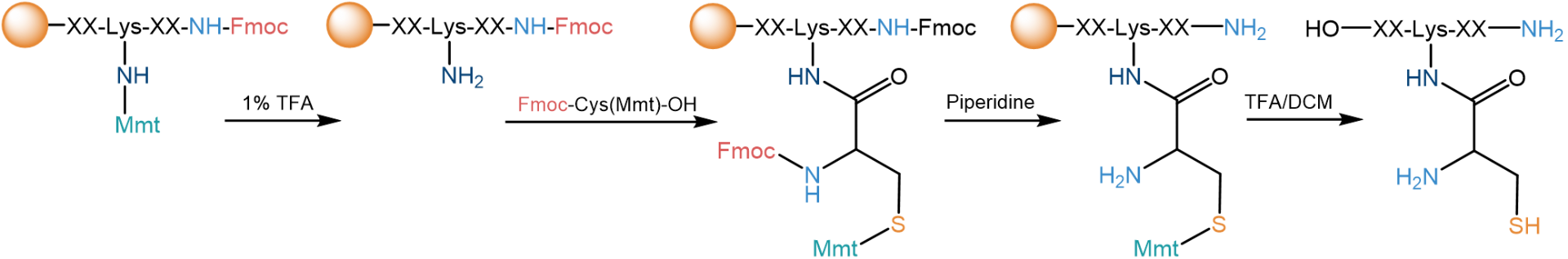
Schematic example for the introduction of an internal 1,2-aminothiol by SPPS into a pentapeptide (XXKXX). An internal lysine, which has been protected with an acid labile monomethoxytrityl (Mmt) group at the side chain amino group, is coupled with a Fmoc/Mmt protected cysteine as the last step of the peptide synthesis.
Besides, 2-cyanobenzothiazoles themselves have a further interesting property: They can react with the active site of cysteine proteases and inactivate these enzymes, although at rather high IC50 values in the µM range.
To make the versatile CBT click reaction accessible to peptide chemists, Iris Biotech has expanded its portfolio with a collection of 2-cyanobenzothiazoles, equipped with various functionalities, e.g., biotin, additional clickable handles (azide, alkyne, DBCO), fluorescent dyes (fluoresceine, indocyanine green), or as single building block (carboxy-and amino-2-CBT). For structures and order codes, please refer to the related products at the bottom of this blog.
→ You are looking for a specifically functionalized 2-CBT derivative not listed in our portfolio? Get in contact for a custom synthesis.
→ Interested in Click Chemistry? Check out our website or download our brochure!
→ You want to know more about ligation technologies? See our booklet!
References:
CBT-Cys click reaction for optical bioimaging in vivo. X. Hu, R. Tang, L. Bai, S. Liu, G. Liang, X. Sun; View. 2023; 20220065. https://doi.org/10.1002/VIW.20220065
Computational study on the mechanism of CBT-Cys click reaction. Y. Xu, C. Qi, C. Wang; CompTheorChem. 2020; 1185: 112874. https://doi.org/10.1016/j.comptc.2020.112874
Biosynthesis of Firefly Luciferin in Adult Lantern: Decarboxylation of ʟ-Cysteine is a Key Step for Benzothiazole Ring Formation in Firefly Luciferin Synthesis; Y. Oba, N. Yoshida, S. Kanie, M. Ojika, S. Inouye; Plos One. 2013; 8: e84023. https://doi.org/10.1371%2Fjournal.pone.0084023
Discovery of selective fragment-sized immunoproteasome inhibitors; L. Kollár, M. Gobec, B. Szilágyi, M. Proj, D. Knez, P. Ábrányi-Balogh, L. Petri, T. Imre, D. Bajusz, G. Ferenczy, S. Gobec, G. Keserű, I. Sosič; Eur J Med Chem. 2021; 219: 113455. https://doi.org/10.1016/j.ejmech.2021.113455
Novel regioselective approach to cyclize phage-displayed peptides in combination with epitope-directed selection to identify a potent neutralizing macrocyclic peptide for SARS-CoV-2; J. Hampton, T. Lalonde, J. Tharp, Y. Kurra, Y. Alugubelli, C. Roundy, G. Hamer, S. Xu, W. Liu; ACS Chem. Biol. 2022; 17: 2911-2922. https://doi.org/10.1021/acschembio.2c00565
Deciphering the cellular targets of bioactive compounds using a chloroalkane capture tag; Ohana, R. Ohana, T. Kirkland, C. Woodroofe, S. Levin, H. Uyeda, P. Otto, R. Hurst, M. Robers, K. Zimmerman, L. Encell, K. Wood. ACS Chem. Biol. 2015; 10: 2316-2324. https://doi.org/10.1021/acschembio.5b00351
Utilizing a simple method for stoichiometric protein Labeling to quantify antibody blockade; R. Ohana, R. Hurst, M. Rosenblatt, S. Levin, T. Machleidt, T. Kirkland, L. Encell, M. Robers, K. Wood; Sci. Rep. 2019; 9: 7046. https://doi.org/10.1038%2Fs41598-019-43469-z
Thiol-cyanobenzothiazole ligation for the efficient preparation of peptide-PNA conjugates; N. Patil, J. Karas, B. Turner, F. Shabanpoor; Bioconjugate Chem. 2019; 30: 73-799. https://doi.org/10.1021/acs.bioconjchem.8b00908
N, S-Double labeling of N-terminal cysteines via an alternative conjugation pathway with 2‑cyanobenzothiazole; W. Wang, J. Gao; Org. Chem. 2020; 85: 1756-1763. https://doi.org/10.1021/acs.joc.9b02959
Mechanistic study of CBT-Cys click reaction and its application for identifying bioactive N-terminal cysteine peptides in amniotic fluid; Z. Zheng, P. Chen, G. Li, Y. Zhu, Z. Shi, Y. Luo, C. Zhao, Z. Fu, X. Cui, C. Ji, F Wang, G. Huang, G. Liang; Chem. Sci. 2017; 8: 214-222. https://doi.org/10.1039/C6SC01461E
A biocompatible condensation reaction for the labeling of terminal cysteine residues on proteins; H. Ren, F. Xiao, K. Zhen, Y.P. Kim, H. Xie, Z. Xia, J. Rao; Angew. Chem. Int. Ed. 2009; 48: 9658-9662. https://doi.org/10.1002/anie.200903627
Genetically encoded 1,2-aminothiols facilitate rapid and site-specific protein labeling via a bio-orthogonal cyanobenzothiazole condensation; D. P. Nguyen, T. Elliott, M. Holt, T. W. Muir, J. W. Chin; J. Am. Chem. Soc. 2011; 133: 11418–11421. https://doi.org/10.1021/ja203111c
Site-specific immobilization of biomolecules by a biocompatible reaction between terminal cysteine and 2-cyanobenzothiazole; P. Wang, C.J. Zhang, G. Chen, Z. Na, S. Q. Yao, H. Sun; Chem. Commun. 2013; 49: 8644-8646. https://doi.org/10.1039/C3CC43566K
Sequence-specific 2-cyanobenzothiazole ligation; C. Ramil, P. An, Z. Yu, Q. Lin; J. Am. Chem. Soc. 2016; 138: 5499–5502. https://doi.org/10.1021/jacs.6b00982
Peptide cleavage induced assembly enables highly sensitive electrochemiluminescence detection of protease activity; M. Cheng, J. Zhuo, X. Zhou, D. Xing; Sens. Actuators B Chem. 2018; 262: 516-21. https://doi.org/10.1016/j.snb.2018.01.191
Injectable hyaluronic acid hydrogels encapsulating drug nanocrystals for long-term treatment of inflammatory arthritis; Y. Gao, D. Vogus, Z. Zhao, W. He, V. Krishnan, J. Kim, Y. Shi, A. Sarode, A. Ukidve, S. Mitragotri; Bioeng. Transl. Med. 2022; 7: e10245. https://doi.org/10.1002/btm2.10245
Affinity bioorthogonal chemistry (ABC) tags for site-selective conjugation, on-resin protein-protein coupling, and purification of protein conjugates; S. L. Scinto, T. R. Reagle, J. M. Fox; Angew. Chem. Int. Ed. 2022; 61: e202207661. https://doi.org/10.1002/anie.202207661
Optimized molecular design of PET probe for the visualization of γ-glutamyltranspeptidase activity in tumors; X. Wang, D. Gao, C. Lu, M. Xie, J. Lin, L. Qiu; New J. Chem. 2022; 46: 10219-10228. https://doi.org/10.1039/D2NJ01688E