Welcome to Iris Biotech
For better service please confirm your country and language we detected.
confirm selection

For better service please confirm your country and language we detected.

Thank you very much for your interest in our products. All prices listed on our website are ex-works, Germany, and may attract customs duties when imported.
You may/will be contacted by the shipping company for additional documentation that may be required by the US Customs for clearance.
We offer you the convenience of buying through a local partner, Peptide Solutions LLC who can import the shipment as well as prepay the customs duties and brokerage on your behalf and provide the convenience of a domestic sale.
Continue to Iris Biotech GmbHSend request to US distributorPublished on 08.06.2016
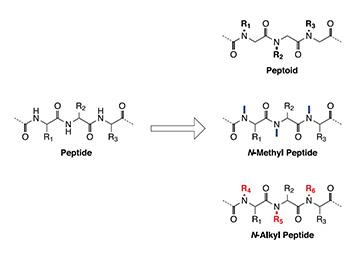
The amide bond between amino acids is the main structural element of peptides. Its facile biological or chemical synthesis in combination with the plethora of available building blocks gives rapid access to a nearly infinite amount of biologically active substances as well as materials with superior and novel properties. However, the peptide bonds lability towards enzymatic degradation and its limited ability to cross biological membranes and barriers are significant disadvantages of peptides that often hinder them in progressing through research and development stages to reach the market.
One way to overcome these drawbacks, which also has been used by nature, while still retaining or even improving the excellent biological activity of peptides, is to alkylate the amide bond. On the one hand, this increases the proteolytic stability towards enzymes by creating a sterically more hindered amide bond. On the other hand, the membrane permeability is improved by substituting the polar amide proton and its ability to form hydrogen bonds by replacing it with an alkyl group. Additionally, N-alkyl groups can also induce better receptor selectivity by increased rigidity and reduced conformational space and can therefore avoid unwanted side-effects.
The term peptoid became broadly applied for oligomers of N-substituted glycines by Bartlett and co-workers in the early 1990s but had already been mentioned earlier. Originally, peptoids had been designed to enable a fast and efficient synthesis with a high diversity of building blocks through a modular approach using simple achiral reagents. Through the modification of the amide bond the enzymatic stability is increased and the missing stereocenter gives the peptoids a greater conformational freedom than the corresponding peptides.
The method of Zuckermann et al. published in 1992 is a fast and reliable way to synthesize peptoids that can be used in automated synthesis. It uses bromoacetic acid (BrAA) as a submonomer which is coupled to the peptoid on solid support at room temperature (RT), using diisopropylcarbodiimide (DIC) and introduces the amine via displacement of the halogen in a rapid two-step reaction cascade. With this method peptoid chains of up to 50 monomers in length can readily be prepared.
Our Peptoid Base Kit contains 24 PP reactors (10 mL, with frit) plus sufficient reagents for 12 syntheses of 15-mer peptoids (100 μmol scale). The Iris Biotech Peptoid Submonomer Kit contains a selection of amines for side chain derivatization (0.1 mol of amine monomers and 0.2 mol of benzylamine).
In contrast to glycine-based peptoids, N-alkylated amino acids containing a sidechain are not as easily available by the previously described submonomer approach because the corresponding bromoacids are significantly more expensive, less reactive and not commercially available in a variety as it is the case for standard Fmoc-amino acids.
One approach to access more complex molecules than oligoglycines is the alkylation of amino acids via their o-nitrobenzenesulfonamide (Ns), due to the mild synthetic conditions and short reaction times that make it compatible with standard solid phase chemistry. Protecting the amine as nitrobenzenesulfonamide facilitates N-alkylation either by using alcohols under Mitsunobu conditions or by using halides, giving access to a vast amount of alkylating agents.
Our Peptide Alkylation Base Kit contains 20 PP-reactors (10 mL, with frit), reagents for Nosyl-protection and -deprotection, as well as reagents for N-alkylation using either Mitsunobu conditions or halide displacement. The Peptide Alkylation Submonomer Kit offers a selection of aliphatic and aromatic alcohols that will allow you to create a variety of N-alkylated peptides via the Mitsunobu reaction.
→ Order our new brochure 'Peptoids & N-Alkylated Peptides' - free of charge! It contains detailed synthetic procedures for the preparation of N-methylated and N-alkylated peptides, as well as further valuable information. Synthesizing N-alkylated peptides has never been so easy!
→ Do you need even more building blocks? In addition to our new kits, Iris Biotech offers a wide variety of amine submonomers as well as alcohol submonomers, and more. Find tables of all available building blocks in our new Peptoids & N-Alkylated Peptides brochure.
References: